Syren's Clinical Trial Control Tower Brings Intelligence to 65+ Active Trials
THE SITUATION
A supply chain that ran on heroics and spreadsheets
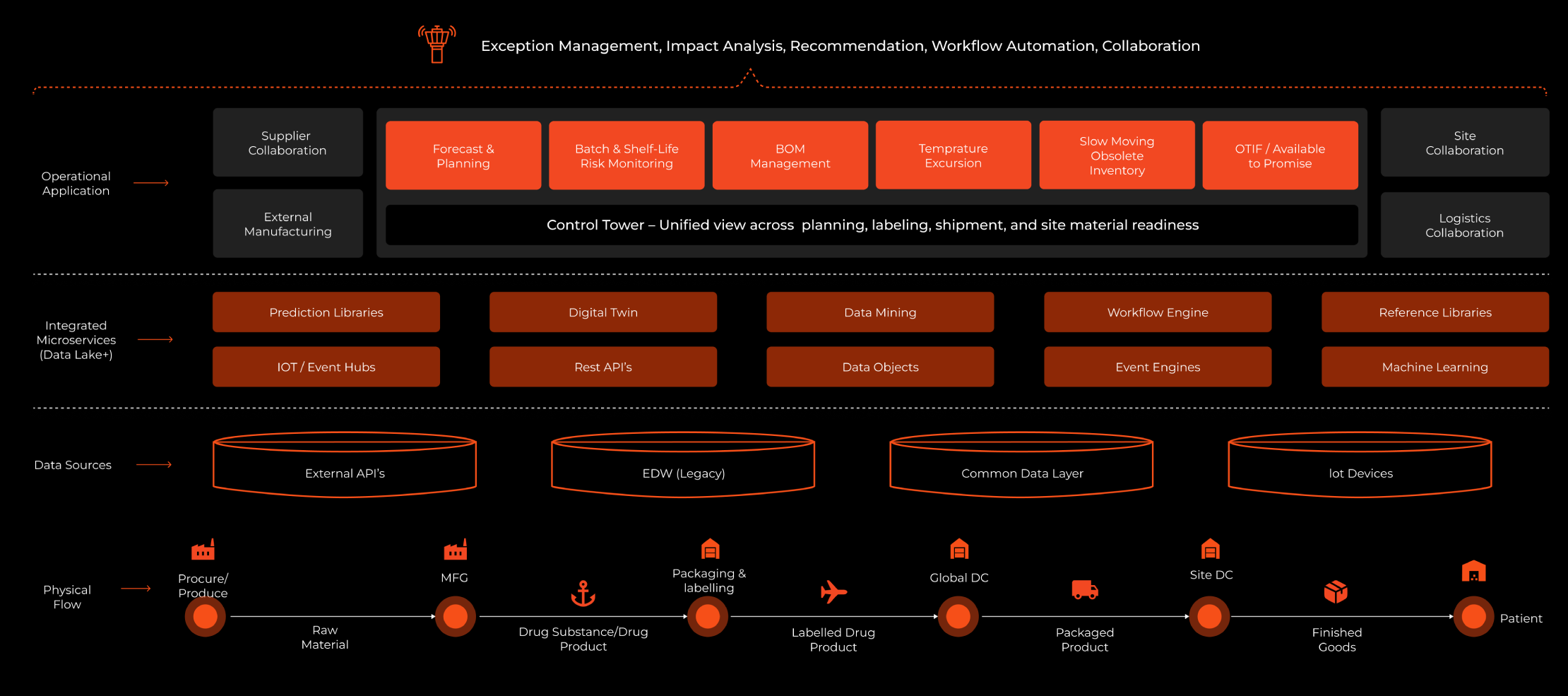
Managing 65+ active clinical trials simultaneously is an extraordinary operational challenge. For this global pharmaceutical company, it meant coordinating demand forecasting, raw material sourcing, drug product manufacturing, packaging, global distribution, and patient-site dispensing; all at once, across multiple geographies, under strict GMP, GCP, and GDP compliance.
Critical data like supply signals, inventory levels, expiry dates, shipment statuses, batch release updates were fragmented across SAP/HANA, WMS, IRT, Veeva Vault, and spreadsheets. There was no unified view, no real-time visibility, and no early warning system.
"By the time we saw the risk in the data, we were already behind. We spent more time finding information than acting on it."
Key Challenges
Six problems that every clinical supply team will recognize.
- Stale Data: Inventory is reported weekly. By the time you see a risk, it's already a crisis. You're reacting — never ahead of it.
- Fragmented Systems: SAP, WMS, IRT, Veeva Vault, and a spreadsheet someone built two years ago. Every decision starts with a data hunt.
- Slow Scenario Planning: Protocol amendments triggered manual reallocation with no fast-modelling capability.
- No Single Source of Truth: Clinical ops, supply chain, and regulatory are each working from different data.
- Manual Expiry Management: Tracking shelf life across 65+ trials, multiple sites, and a global depot network manually was a constant drain.
- Late Shipment Alerts: Temperature excursions, lane deviations, delayed batches, the alerts come after the damage is done.
The Solution
Syren built a centralized Clinical Trial Control Tower, a centralized command center aggregating all data sources with an intelligence layer that surfaces risks and recommends actions in real time.
| What We Built | How it Works |
|---|---|
| End-to-End Tracking | Raw material to patient |
| Unified Data Model | Consolidated into a single data model on AWS |
| Intelligence Built In | Pre-integrated intelligence |
| Role-based Dashboards | Each user sees exactly what they need |
| Closed Loop. Self-Healing. | Routine exceptions resolved automatically with audit trails |
Key Capabilities
Real-Time Inventory Monitoring
Live site-level inventory with automated risk flags.
Packaging & Labelling Management
Batch release and labelling status tracked by country, end-to-end.
Study Grading & Enrollment Tracking
Integrates with trial management tools to flag enrollment changes before they ripple downstream.
Shipment & Temperature Monitoring
Real-time tracking of documentation, temperature data, and cold-chain integrity.
KPI Dashboards & Reporting
Role-based views covering patient impact, expiry risk, and inventory health.
"Scenario planning used to take days. Now we can model a protocol amendment and see the supply impact within the hour."
Business Impact
The deployment of the Clinical Trial Supply Chain Control Tower delivered measurable, multi-dimensional value:
Conclusion
Clinical supply chains have always been complex. Protocols change. Sites go on hold. Expiry dates don't reverse. What changes with the Control Tower is not the complexity; it's who's in control of it.
By consolidating every data source into a single intelligent platform, backed by Digital Twin modelling, ML-powered analytics, IoT-event-driven alerting, and closed-loop write-backs; the Control Tower gives clinical supply teams the full picture, in real time, with the tools to act on it immediately.
The system gets smarter as execution data accumulates. It scales as new trials are added. And it provides the compliance foundation: audit trails, approval workflows, self-healing automation, to meet regulatory requirements as they evolve.
The right drug. The right patient. The right time. It's the only outcome that matters.